
Johnson & Johnson's Ethicon Withdraws Physiomesh
This is Michael Brady Lynch, Lead Trial Attorney for The Michael Brady Lynch Firm. Every year, surgeons perform around 150,000 ventral hernia repairs in the United States. Doctors can perform the operation laparoscopically or by opening the site surgically. During surgery, it is very common for surgeons to stitch a mesh patch over a hernia
Read More
The Science Behind Viagra and Melanoma
In 2014, JAMA Internal Medicine published a scientific study finding that men using Viagra have an increased risk of developing the deadly skin cancer, melanoma. Since the FDA approved Viagra in 1998, 45 million men taking the drug may have unknowingly doubled their risk of developing melanoma. In the United States, 10,000 people die from
Read MoreFDA Changes Testosterone Product Labeling
This is Michael Brady Lynch, Lead Trial Attorney for The Michael Brady Lynch Firm. The FDA has approved new labeling for all prescription testosterone products. The new warning will alert prescribers to the abuse potential of testosterone along. Additionally, it will detail several serious side effects from using this class of drug. These side effects
Read More
Xarelto Purposely Pushed to Market Regardless of Dangers
This is Michael Brady Lynch, Lead Trial Attorney for The Michael Brady Lynch Firm. The truth is coming to light as reports surface which proves Xarelto manufacturers, Johnson & Johnson and Bayer knew the deadly blood-thinner was too dangerous to be on the market. Yet, they did nothing to protect the American consumer. Instead, they
Read More
FDA Approves New Invokana Drug Amid Lawsuits
This is Michael Brady Lynch, lead trial attorney for The Michael Brady Lynch Firm. The FDA is not taking the deadly side effects of Invokana seriously. Instead, they approved a new version of the drug. Janssen Pharmaceuticals designed another SGLT2 inhibitor, Invokamet XR to treat Type 2 diabetes. Invokamet XR combines deadly Invokana and metformin
Read More
Invokana Manufacturer Looking for Type 1 Diabetes Approval
Invokana is projected to make billions of dollars in sales as the obesity and diabetes market will double by 2022. Invokana is already the most prescribed Type 2 diabetes drug in its class. Now, manufacturer Janssen Pharmaceuticals is pushing for approval for Type 1 diabetes also. This is troubling since Type 1 and Type 2
Read More
FDA in No Rush to Prove Essure Risks
After 10,000 reports of harmful side effects from Essure, the FDA mandated that Bayer, the Essure manufacturer needs to perform a new study concentrating on the harmful side of effects of the device. Lawmakers and the public are outraged that not only has Bayer finally submitted its proposal for the study almost a year later,
Read More
Michael Brady Lynch Now Executive Committee Member of “STEP”
Lead trial attorney and firm founder, Michael Brady Lynch has been appointed as an Executive Committee Member of the American Association for Justice’s Section on Toxic, Environmental and Pharmaceutical Torts (STEP). This prestigious position allows Attorney Michael Brady Lynch to continue to be a driving force in many nationwide litigations. The American Association for Justice
Read More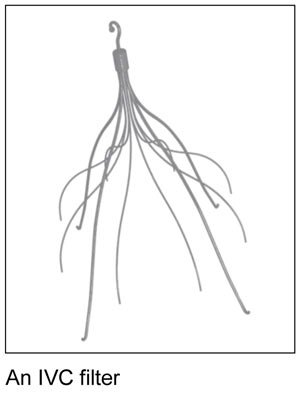
Judge Selects IVC Filter Bellwether Cases for 2017 Trial
This is Michael Brady Lynch, lead trial attorney for The Michael Brady Lynch Firm. There is forward momentum in IVC filter cases. The U.S. District Judge Richard L. Young is overseeing the pretrial process for the Cook IVC filter lawsuits. Judge Young announced the three claims selected for bellwether trials set to begin in 2017.
Read More
Defective Medical Device May Lead to Deadly Bleeds and Death
This is Michael Brady Lynch, lead trial attorney for The Michael Brady Lynch Firm. When a patient is taking Coumadin, a blood thinning drug, a medical professional must closely monitor a patient. This is to ensure they aren’t developing deadly blood clots. For many, they rely on in home devices instead of daily trips to
Read More