
Xarelto Bellwether Trial Begins in New Orleans
The multi-district litigation (MDL) for Xarelto in the U.S. District Court, Eastern District of Louisiana is underway. Right now, attorneys are presenting the first of four Xarelto Bellwether trials to a New Orleans jury in hopes that they will hold manufacturer Johnson and Johson along with Bayer accountable for Xarelto’s fatal side effects. First Xarelto
Read MoreFDA Approves Dangerous Onglyza Cocktail
The FDA has approved a once-daily oral fixed dose medication called Qtern. Qtern combines Onglyza, a DPP-4 inhibitor and an SGLT2 inhibitor called Farxiga. Qtern is for the treatment of Type 2 diabetes. DDP-4 inhibitors like Onglyza are being linked to heart failure while SGLT2 inhibitors like Invokana are being linked to severe kidney damage.
Read More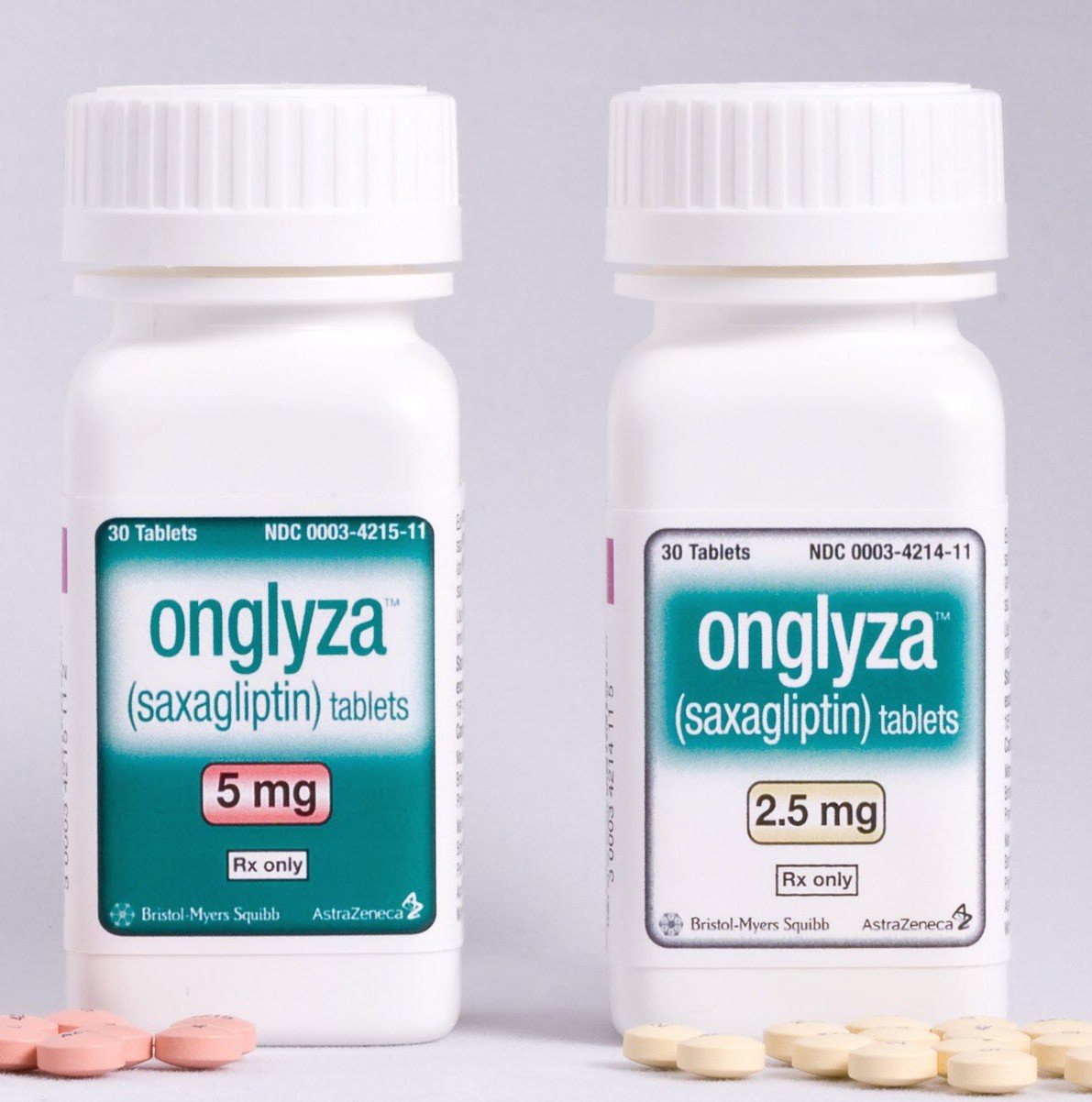
Onglyza Manufacturer Likely Knew Heart Failure Risk Before Approval
Almost 10% of the U.S. population has Type 2 diabetes. This means big bucks for pharmaceutical companies like AstraZeneca. They knew if they could develop a medication to treat Type 2 diabetes, then they could cash in. Expected annual sales of these drugs will reach $2.47 billion by 2018. Plaintiffs allege that AstraZeneca knew their
Read More
Is Invokana Worth the Risk?
If consumers knew every side effect before taking a drug, would they still choose to take it? What people are unaware of is that they aren’t being told the side effects. Manufacturers conceal them or the FDA identifies potential risks but approves the drugs anyway. This happened with the Type 2 diabetes medication, Invokana. What
Read More
Hundreds of Fosamax Fracture Cases Reinstated
This week the Third Circuit Court of Appeals decided to reverse a ruling by U.S. District court Joel Pisano. Judge Pisano dismissed all federal court Fosamax cases on federal preemption grounds. The Third Court decided that whether the FDA would have rejected a Fosamax label change by the manufacturer, Merck to warn consumers about an
Read More
IVC Filter Lawsuits Mounting
Around 2,000 IVC filter lawsuits are now pending against manufacturer C.R. Bard in a multidistrict ligation (MDL No.2641). This MDL is located in the U.S. District Court for the District of Arizona. It surrounds consumers being injured by serious design flaws caused by the filter. IVC Filters are supposed to prevent deadly blood clots from
Read More
The Michael Brady Lynch Firm Releases Four Part Expose on Taxotere
Thousands of cancer survivors along with Winter Park-based law firm, The Michael Brady Lynch Firm are teaming up to fight against Sanofi-Aventis, the manufacturer of the common chemotherapy drug, Taxotere. Sanofi knew Taxotere brought terrible side effects to cancer survivors. Instead of informing the public, they made strides to hide the results to their American
Read More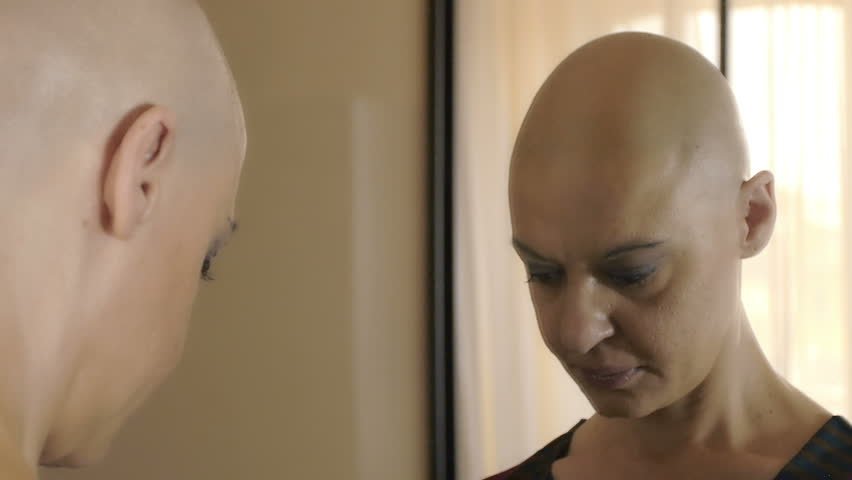
Safer Alternatives to Taxotere
Taxotere manufacturer, Sanofi-Aventis knew that their chemotherapy drug could cause permanent hair loss back in the 1990s. The company disclosed to other countries this side effect, but they hid it from patients in the United States. Instead of protecting consumers, they protected their bottom line, especially knowing there are safer alternatives on the market.
Read More