
FDA Reports 30 Deaths and 10,000 Injuries From Essure
The FDA recently updated their ongoing safety review of the birth control device, Essure. In the last two years, the FDA received over 10,000 reports of injuries and 30 deaths from the device. This is just the small amount actually reported to the agency. Statistics have shown that many neglect to report adverse events to
Read More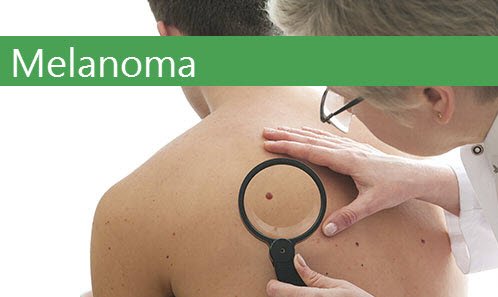
Researchers Find Melanoma Causing Cell – Implications for Viagra Melanoma Victims
Skin cancer is nothing to take lightly. It is the most common form of cancer in the United States. Melanoma being the deadliest. It begins by invading the melanocytes (pigment-producing cells). The melanocytes are responsible for mole formation. Melanoma will develop from these moles and change the color pigmentation. If caught early with regular skin
Read More
New Study Suggests Xarelto Causes More Bleeding Than Even Pradaxa
A study published in JAMA Internal Medicine suggests Xarelto causes more bleeding than Pradaxa. Researchers found that people using Xarelto may be more likely to have a serious bleeding event than those taking Pradaxa. This could explain why manufacturer, Johnson & Johnson is facing close to 20,000 Xarelto lawsuits. JAMA Study: Xarelto Causes More Bleeding Researchers
Read More
FDA Recalls around 30,000 Abbott-Thoratec’s HeartMate II LVAS Devices
The FDA issued a Class I Recall against HeartMate II Left Ventricle Assist (LVAS) Pocket System Controller manufacturer, Abbott-Thoratec. This is the most serious formal recall since the issue with the device can result in serious injury or death. Regulators are concerned that when switching to the backup system the device can malfunction and thus
Read MoreJohnson & Johnson Shows Zero Remorse
This week Johnson & Johnson petitioned a Missouri state appeals court to dismiss a $72 million verdict. Last year, a jury awarded the settlement to the family of an Alabama woman who claimed the company’s talcum-based hygiene product caused her ovarian cancer. The company argued to a three-judge panel that a lawsuit filed by an
Read More
Invokana Litigation Heading to Trial
Those who trusted the Type 2 diabetes drug, Invokana or Invokamet, but ended up with the deadly side effect of diabetic ketoacidosis are getting their time in court. According to a Case Management Order dated May 1st, the U.S. District Court of New Jersey established a discovery schedule. This schedule will address the selection of
Read More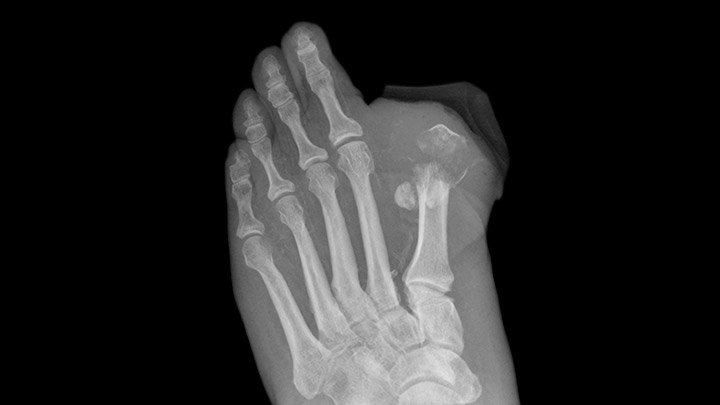
FDA Gives Invokana Black Box Amputation Warning
Based on new data from two large clinical trials, the FDA issued a new Black Box warning for the Type 2 diabetes drug, Invokana, Invokamet and Invokamet XR. Both studies the organization reviewed showed an increased risk of leg and foot amputations to patients using Invokana. Because of this, the FDA required Invokana to have
Read More
FDA Updates Fluoroquinolone Warning
After an FDA safety review, the organization approves new safety labeling for the antibiotic, fluoroquinolone. Recently, published studies say the drug may result in the detachment of the retina in the eyes, bulges or tears in the aorta blood vessel called aortic aneurysm or aortic dissection. The updated warning will include retina issues. About Fluoroquinolone
Read More
Johnson & Johnson & Their Multimillion-Dollar Lawsuits
Currently, there are multiple trials against Johnson & Johnson for many of its medications and products. These products are severely injuring consumers. The main ones are Transvaginal Mesh, Talcum Powder, Pinnacle Hip Implants, Risperdal, Invokana, and Power Morcellators. Right now, tens of thousands of cases are pending all over the U.S. against them. Despite this
Read More
Onglyza Litigation Update
At The Michael Brady Lynch Firm, we work tirelessly to ensure you receive the most compensation for your injury. Right now, we are working hard to ensure our clients, who experienced heart failure from taking Onglyza, are going to receive their justice in court. The Onglyza litigation is starting to take shape. Onglyza Litigation –
Read More