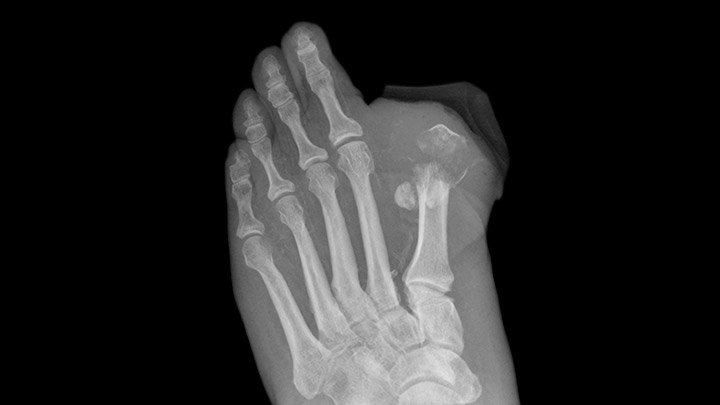
FDA Gives Invokana Black Box Amputation Warning
Based on new data from two large clinical trials, the FDA issued a new Black Box warning for the Type 2 diabetes drug, Invokana, Invokamet and Invokamet XR. Both studies the organization reviewed showed an increased risk of leg and foot amputations to patients using Invokana. Because of this, the FDA required Invokana to have
Read More
FDA Updates Fluoroquinolone Warning
After an FDA safety review, the organization approves new safety labeling for the antibiotic, fluoroquinolone. Recently, published studies say the drug may result in the detachment of the retina in the eyes, bulges or tears in the aorta blood vessel called aortic aneurysm or aortic dissection. The updated warning will include retina issues. About Fluoroquinolone
Read More
Johnson & Johnson & Their Multimillion-Dollar Lawsuits
Currently, there are multiple trials against Johnson & Johnson for many of its medications and products. These products are severely injuring consumers. The main ones are Transvaginal Mesh, Talcum Powder, Pinnacle Hip Implants, Risperdal, Invokana, and Power Morcellators. Right now, tens of thousands of cases are pending all over the U.S. against them. Despite this
Read More
Onglyza Litigation Update
At The Michael Brady Lynch Firm, we work tirelessly to ensure you receive the most compensation for your injury. Right now, we are working hard to ensure our clients, who experienced heart failure from taking Onglyza, are going to receive their justice in court. The Onglyza litigation is starting to take shape. Onglyza Litigation –
Read More
Invokana Sales Decline as Lawsuits Increase
Slowly, Type 2 diabetics and their families are becoming more aware of the dangers of SGLT2 inhibitors like Invokana. Recently, Johnson & Johnson, the manufacturer of Invokana released their 2017 First-Quarter Results. U.S. sales of Invokana fell by $50 million, which is almost 20%. Dangers of Invokana Invokana is the brand name of the drug canagliflozin,
Read More
Express Scripts Drops Onglyza Coverage over Safety Concerns
It should be troubling to all users of Onglyza and Kombiglyze XR that the largest pharmacy benefits manager in the United States, Express Scripts has dropped the Type 2 Diabetes medication. Express Scripts stated that the drug’s side effects were too concerning. Why is a corporation refusing to injury people, yet the FDA still won’t
Read More
Xarelto Trial Update: Week One Recap
The first week of the Xarelto trial (MDL No. 2592) in the U.S. District Court, Eastern District of Louisiana was a great first step for our clients to receive justice. Plaintiffs rested on Friday, and this week centers on the defense. About the Xarelto Trial Xarelto has been significantly responsible for numerous deaths and horrible side
Read More
Xarelto Bellwether Trial Begins in New Orleans
The multi-district litigation (MDL) for Xarelto in the U.S. District Court, Eastern District of Louisiana is underway. Right now, attorneys are presenting the first of four Xarelto Bellwether trials to a New Orleans jury in hopes that they will hold manufacturer Johnson and Johson along with Bayer accountable for Xarelto’s fatal side effects. First Xarelto
Read MoreFDA Approves Dangerous Onglyza Cocktail
The FDA has approved a once-daily oral fixed dose medication called Qtern. Qtern combines Onglyza, a DPP-4 inhibitor and an SGLT2 inhibitor called Farxiga. Qtern is for the treatment of Type 2 diabetes. DDP-4 inhibitors like Onglyza are being linked to heart failure while SGLT2 inhibitors like Invokana are being linked to severe kidney damage.
Read More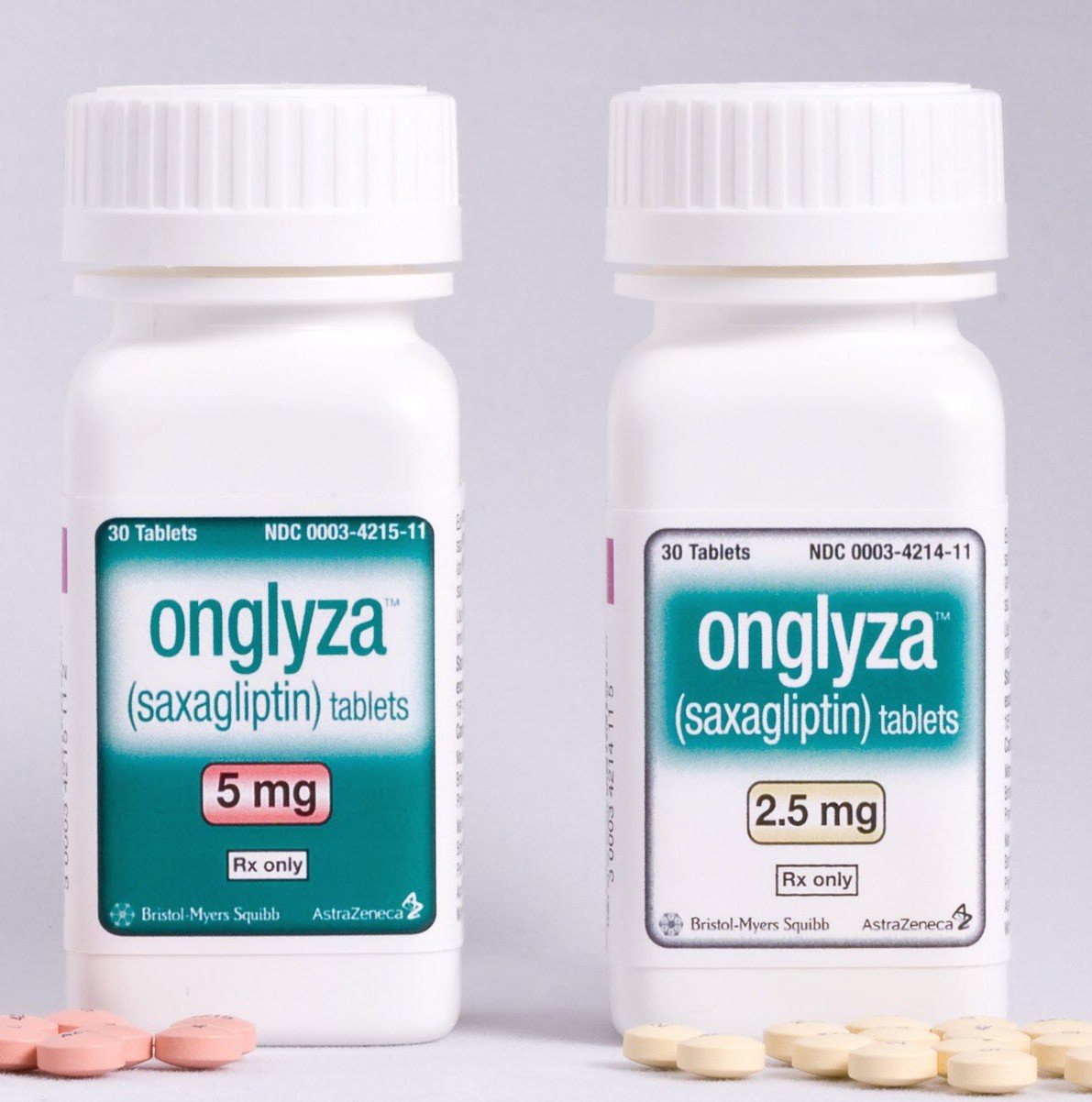
Onglyza Manufacturer Likely Knew Heart Failure Risk Before Approval
Almost 10% of the U.S. population has Type 2 diabetes. This means big bucks for pharmaceutical companies like AstraZeneca. They knew if they could develop a medication to treat Type 2 diabetes, then they could cash in. Expected annual sales of these drugs will reach $2.47 billion by 2018. Plaintiffs allege that AstraZeneca knew their
Read More