Actemra & Heart Attack
Studies have linked the rheumatoid arthritis drug Actemra (tocilizumab injection) to serious side effects including heart failure, stroke, interstitial lung disease, and more than 1,100 deaths. About Actemra In early 2010, the FDA first approved the Roche manufactured drug, Actemra to treat moderate to severe rheumatoid arthritis (RA). The drug works by reducing inflammation.
Read MoreActemra & Stroke
Studies have linked the rheumatoid arthritis drug Actemra (tocilizumab injection) to serious side effects including stroke, interstitial lung disease, heart attack and more than 1,100 deaths. About Actemra In early 2010, the FDA first approved the Roche manufactured drug, Actemra to treat moderate to severe rheumatoid arthritis (RA). The drug works by reducing inflammation. Over
Read MoreActemra & Bone Marrow Failure
Studies have linked the rheumatoid arthritis drug Actemra (tocilizumab injection) to serious side effects including bone marrow failure and also to disorders like hematophagic histiocytosis, stroke, heart attack and more than 1,100 deaths. About Actemra In early 2010, the FDA first approved the Roche manufactured drug, Actemra to treat moderate to severe rheumatoid arthritis (RA). The
Read MoreActemra & Pancreatitis
Studies have linked the rheumatoid arthritis drug Actemra (tocilizumab injection) to serious side effects including pancreatitis, stroke, heart attack and more than 1,100 deaths. About Actemra In early 2010, the FDA first approved the Roche manufactured drug, Actemra to treat moderate to severe rheumatoid arthritis (RA). The drug works by reducing inflammation. Over 1.5 million Americans
Read More
Actemra & Acute Respiratory Distress Syndrome
Studies have linked the rheumatoid arthritis drug Actemra (tocilizumab injection) to serious side effects including Acute Respiratory Distress Syndrome, stroke, heart attack and more than 1,100 deaths. This comes just a mere 6 years after the FDA approved the medication. About Actemra In early 2010, the FDA first approved the Roche manufactured drug, Actemra to treat moderate
Read More
FDA Connected to Manufacturer of Deadly Arthritis Drug Actemra
Introduced in 2010, Actemra treats rheumatoid arthritis. Unlike other drugs on the market, it was supposed to not carry the same potentially deadly cardiovascular and lung complications. However, according to a new report from STAT, Actemra is not as safe as FDA and manufacturer, Roche would want consumers to believe. In fact, the STAT report suggests
Read More
Deadly Side Effects of Actemra
An independent research group called STAT analyzed a half million adverse reports from patients using the Rheumatoid Arthritis (RA) drugs. They uncovered that over 1,000 people who used Actemra died from heart attacks, stroke, and lung complications. Other RA medications warn of these side effects, but Actemra does not. Actemra & Heart Attacks Just six years after approval,
Read More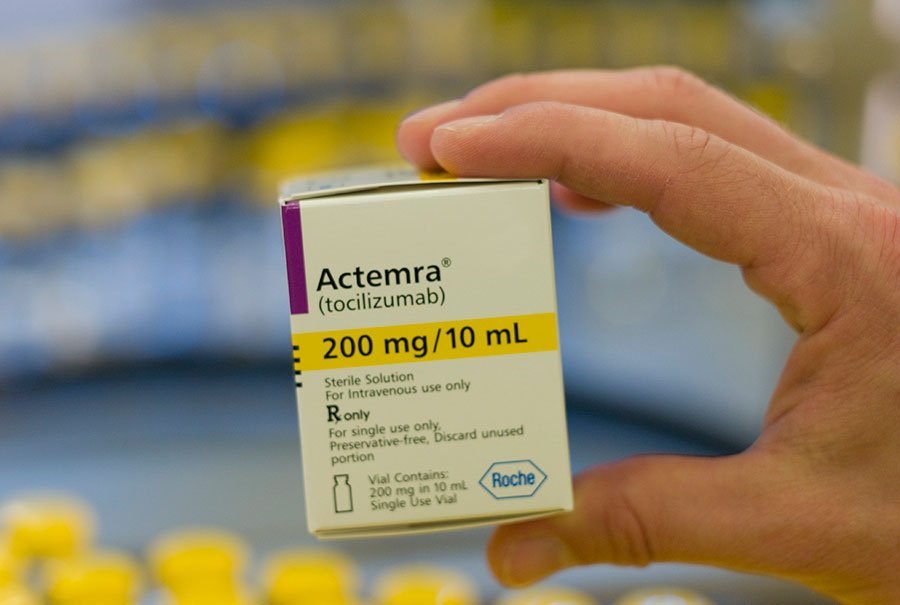
New Study Finds Popular RA Drug Actemra Is Deadly
After analyzing over 500,000 reports of adverse events experienced from Rheumatoid Arthritis (RA) drugs, STAT found that over 1,000 people who used Actemra died from heart attacks, stroke, heart failure and lung complications. Other RA medications warn of these side effects, but Actemra does not. About Actemra Over 1.5 million Americans have rheumatoid arthritis,
Read More