
Invokana Lawsuits Grow
In the past month, there have been almost 100 new cases filed in the U.S. District Court, District of New Jersey alone. These cases join the nearly 800 ones still pending there. These numbers are expecting to grow even more in the coming months as more people are injured from the Type 2 diabetes medication.
Read More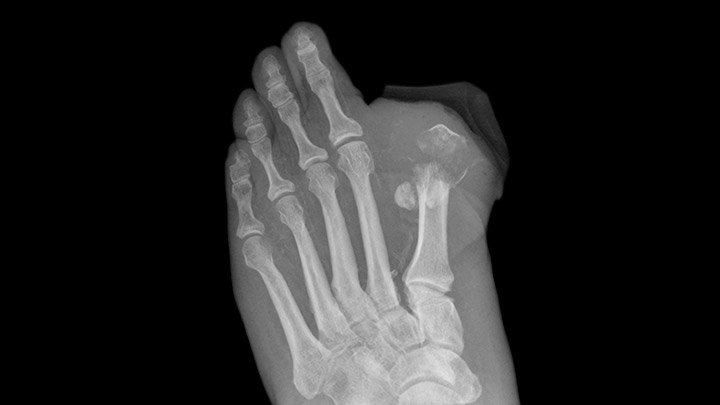
Invokana Users Twice as Likely to Experience Amputation
This summer results from two clinical trials prompted the FDA to warn that popular type 2 diabetes medication, Invokana causes an increased risk for leg and foot amputation. Why the Danger Canagliflozin, the active ingredient in Invokana, lowers blood sugar levels in those with type 2 diabetes. It works by increasing the rate of excess
Read More
Invokana Lawsuit Schedules Science Day for July
According to a Case Management Order, the U.S. District Court, District of New Jersey will convene Science Day on July 19, 2017, at 9:30 a.m. State court judges presiding over other this Invokana lawsuit and others will be invited to attend. Science Day Science Day is a common proceeding in large, complex litigations. On this
Read More
The Deadly Side Effects of Invokana
Invokana was the first in a new class of drug for Type 2 diabetics. It was supposed to help people easily manage their diabetes without insulin by lowering blood sugar by causing the kidneys to remove sugar from the body via urine. This prevents blood from reabsorbing the sugar. In March 2013, the FDA approved
Read More
FDA Gives Invokana Black Box Amputation Warning
Based on new data from two large clinical trials, the FDA issued a new Black Box warning for the Type 2 diabetes drug, Invokana, Invokamet and Invokamet XR. Both studies the organization reviewed showed an increased risk of leg and foot amputations to patients using Invokana. Because of this, the FDA required Invokana to have
Read More
Invokana Sales Decline as Lawsuits Increase
Slowly, Type 2 diabetics and their families are becoming more aware of the dangers of SGLT2 inhibitors like Invokana. Recently, Johnson & Johnson, the manufacturer of Invokana released their 2017 First-Quarter Results. U.S. sales of Invokana fell by $50 million, which is almost 20%. Dangers of Invokana Invokana is the brand name of the drug canagliflozin,
Read More
Is Invokana Worth the Risk?
If consumers knew every side effect before taking a drug, would they still choose to take it? What people are unaware of is that they aren’t being told the side effects. Manufacturers conceal them or the FDA identifies potential risks but approves the drugs anyway. This happened with the Type 2 diabetes medication, Invokana. What
Read More
FDA Approves New Invokana Drug Amid Lawsuits
This is Michael Brady Lynch, lead trial attorney for The Michael Brady Lynch Firm. The FDA is not taking the deadly side effects of Invokana seriously. Instead, they approved a new version of the drug. Janssen Pharmaceuticals designed another SGLT2 inhibitor, Invokamet XR to treat Type 2 diabetes. Invokamet XR combines deadly Invokana and metformin
Read More
Invokana Manufacturer Looking for Type 1 Diabetes Approval
Invokana is projected to make billions of dollars in sales as the obesity and diabetes market will double by 2022. Invokana is already the most prescribed Type 2 diabetes drug in its class. Now, manufacturer Janssen Pharmaceuticals is pushing for approval for Type 1 diabetes also. This is troubling since Type 1 and Type 2
Read More
FDA Stengthens Invokana’s Label to Reflect Severe Kidney Damage
The FDA just revised Invokana’s labeling to warn of acute kidney damage when taking canagliflozin, the active ingredient in the Type 2 Diabetes drug. The label changes come on the heels of surfacing reports. These reports state that since Invokana’s approval in March 2013, the FDA received 101 confirmed cases of acute kidney damage. These
Read More